In any section where the clause is applicable to your device, but it is a function your firm is outsourcing, then you will need to state this in the manual and you will need to reference the supplier quality management procedure (i.e. If you are only purchasing our quality manual template, then you will need to substitute your own procedure numbers for the numbers we used. In order to address sub-clause 4.2.2b), we included references to our standard operating procedures in each section of the manual where the procedure requirement is found. You also delete the draft rationale for non-applicability from the appropriate section of the manual. If the sub-clause is applicable, then you delete the sub-clause from the list of sub-clauses that are not applicable. If the sub-clause is not applicable to your device, then you delete that section of the manual’s content and change the green text to the black font. 
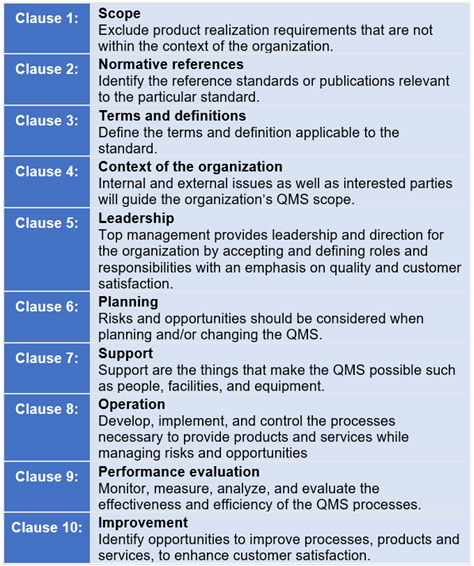
In both places, this content is in green font. In addition, in the sections of the manual where those sub-clauses are found, we have inserted a draft rationale for non-applicability. In order to address sub-clause 4.2.2a), we included a list of the most common sub-clauses that are identified as non-applicable. Finally, the fourth requirement is that the quality manual shall outline the structure of the documentation used in the quality management system. Third, a description of the interaction between the processes of the quality management system. Second, t he quality manual must include documented procedures for the quality management system or reference to them. However, in ISO 13485 it is only permitted to exclude design controls, and sub-clauses within clauses 6, 7, and 8 may be identified as non-applicable depending upon the nature of the products or services. In ISO 9001:2015, any clause may be excluded. First, you must define the scope of the quality management system, including details of and justification for any exclusion or non-application. There are four requirements for the content of a quality manual in that sub-clause. It is the primary document meeting the applicable regulatory requirements for a Quality Manual as defined in clause 4.2.2 of ISO 13485:2016. POL-001 is Medical Device Academy’s template for a quality manual. This requirement is found in clause 4.2.1(b). Once you have met the ISO 13485:2016 requirements and are granted certification you can then advertise your certification to ISO 13485:2016.Unlike ISO 9001:2015, the ISO 13485:2016 standard requires that manufacturers establish a quality manual. In fact, the United States FDA made a formal announcement that they will be replacing parts of the existing Quality System Regulation (QSR), with the international standard: ISO 13485:2016. Additionally, ISO:13485:2016 has many common requirements to address FDA 21 CFR Part 820. If you are interested in an integrated management system, learn more about how to harmonize the two by comparing ISO 13485:2016 with ISO 9001:2015. ISO 13485:2016 = ISO 9001:2008 + additional requirements specific to the medical device industry, which is arranged in an 8 clause format (whereas ISO 9001:2015 is based on a 10 clause format). To successfully implement ISO 1485:2016 within your organization, you must satisfy the requirements within clauses 4-8, along with meeting customer and applicable statutory and regulatory requirements. The requirements to be applied to your quality management system (QMS) are covered in clauses 4-8. The ISO 13485 requirements encompass 8 clauses with supporting subclauses. ISO 13485:2016 is the latest version of ISO 13485. Here we have broken down the ISO 13485:2016 requirements of the standard to make it easier to understand and put into action.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
